Dr. Jim Henry, P.E. Professor of Engineering University of Tennessee at Chattanooga 615 McCallie Avenue Chattanooga, TN Dr.
|
|
|
- Hugh Dickerson
- 6 years ago
- Views:
Transcription
1 Aubrey Gunter Green Team - Distillation College of Engineering and Computer Science University of Tennessee at Chattanooga 615 McCallie Avenue Chattanooga, TN To: Dr. Jim Henry, P.E. Professor of Engineering University of Tennessee at Chattanooga 615 McCallie Avenue Chattanooga, TN Dr. Henry: The following report provides a detailed account of the Green Team s experimental work and all other relevant information related to their study of the distillation column at the University of Tennessee at Chattanooga located in Grote room 115. Aubrey Gunter Green Team Senior Undergraduate Student Chemical and Environmental Engineering University of Tennessee at Chattanooga
2 Distillation Column Experiments, Batch and Continuous University of Tennessee at Chattanooga College of Engineering and Computer Science Engineering 435 Chemical Process Laboratory Section 001 Author: Aubrey Murray Gunter Co-Authors: Alok Patel, Paul Pearman To: Dr. Jim Henry Cc: Dr. Frank Jones October 24, 2002
3 ABSTRACT: A distillation column was studied at the University of Tennessee at Chattanooga, School of Engineering and Computer Science for the binary system of methanol and water. The purpose of this study of the distillation column was to determine the composition of methanol in the distillate collected for various reflux percentages under conditions of continuous and batch distillation. Two methods were used to determine the composition of the distillate, density and temperature. The distillate flow rate for various reflux percentages under conditions of continuous and batch distillation was also studied. Two methods were used to study the distillate flow rate. The first method was performed by manually observing the collected distillate level over time. The second method involved a flow meter and the LabVIEW program that calculates the flow rate. The composition of each tray for various reflux percentages under conditions of continuous and batch distillation was also studied. The temperature method was used to study this tray composition. An energy study was also performed on the condenser. The relationship between the manual study and computer study of distillate flow rate is given in the following equation: Q Manual = (1.68±1.08)Q LabVIEW - 188(±11.5), where Q is the flow rate is L/min. The methanol composition of the distillate and for each tray was found to have a proportional relationship to the reflux percentage for both batch distillation and continuous distillation experiments. The energy study revealed that the energy removed by the condenser is higher for batch distillation than for continuous distillation.
4 TABLE OF CONTENTS: Page Introduction x Theory x Equipment..x Procedure x Results x Discussion of Results..x Conclusions.x Recommendations...x Nomenclature..x References...x Appendices..x
5 INTRODUCON: The purpose of this study of the distillation column by the Green Team was to determine the composition of the distillate collected for various reflux percentages under conditions of continuous and batch distillation. Two methods were used to determine the composition of the distillate, density and temperature. The Green Team also studied the distillate flow rate for various reflux percentages under conditions of continuous and batch distillation. Two methods were used to study the distillate flow rate. The first method was performed by manually observing the collected distillate level over time. The second method involved a flow meter and the LabVIEW program that calculates the flow rate. The composition of each tray for various reflux percentages under conditions of continuous and batch distillation was also studied. The temperature method was used to study this tray composition. The Green Team also performed an energy study on the condenser. Binary distillation is the process in which a mixture of 2 chemicals are separated based upon each chemical s vapor pressure, or how easily each chemical vaporizes. For the purposes of this experiment, only a binary mixture of methanol and water was studied. When two pure chemicals are mixed and heated to boiling, the chemical with the lowest vapor pressure, i.e. the lowest boiling point, will vaporize first. This component of the binary mixture is called the light component, and commonly referred to as component A the mixture. The other component of the binary mixture is called the heavy component, and is commonly referred to as component B of the mixture. A distillation column is a column composed of a series of trays stacked vertically, with a condenser, reboiler, and a feed. The feed is the mixture of chemicals, in this case methanol and water, which is fed into the middle section of the distillation column. The reboiler provides energy to the distillation column in the form of heat to cause the light component, component A, of the binary mixture to vaporize first. The reboiler is located at the bottom of the distillation column. The product stream of the reboiler is known as the bottoms product. The condenser is located at the top of the distillation column and removes energy from the distillation column. The purpose of the condenser is to condense the vapor leaving the top tray of the column. Reflux percent is the percent of liquid condensed in the condenser that is allowed to be collected as distillate. The distillate is the product stream from the top of the distillation column, which is rich in component A of the binary mixture. The remaining liquid that is not collected as distillate, termed reflux, is returned to the distillation column for further separation. A distillation column can be run via batch operation or continuous operation. In batch distillation, a batch of chemicals, in this case methanol and water, is first fed into the column and the liquid part of this mixture settles in the reboiler. Once the reboiler is filled with the desired finite batch of the binary mixture, energy is added to the reboiler in the form of heat to start the experiment. In continuous distillation, the feed enters the column continuously. Heat is constantly supplied to the reboiler, and a continuous flow of distillate and bottoms product is collected or returned to the feed tank.
6 THEORY: The composition of the distillate collected was analyzed in two ways. One method for determining composition is density. This calculated density was then compared with a tabulated list of densities located in Perry s Handbook. To find the density, a measured volume of collected distillate was weighed. The formula for density is given below. Density = Mass/volume (1) The second method for determining composition of the distillate collected, and the composition for each tray, is temperature. Using a vapor-liquid equilibrium diagram from tabulated data in Perry s Handbook for methanol and water mixture, the known temperature of the liquid in each tray can be used to find the composition of the liquid and vapor. The following diagram in Figure 1 illustrates this method Perry's Data for Methanol-Water Vapor Equilibrium Line Data x Data y 90.0 Temperature, oc Liquid Equilibrium Line x,y Figure 1. Vapor-Liquid Equilibrium Diagram for Methanol and Water For example, at a temperature of 78.0 o C, the composition of liquid of the light component, in this case methanol, is 0.30 mole fraction, and the composition of methanol in the vapor is 0.67 mole fraction.
7 Reflux percentage is the percent of liquid condensed in the condenser that is allowed to be collected as distillate. The distillate is the product stream from the top of the distillation column, which is rich in component A of the binary mixture. The remaining liquid that is not collected as distillate, termed reflux, is returned to the distillation column for further separation. At 95% reflux, 5% of the condensed liquid from the condenser is collected as distillate. The remaining 95% of the condensed liquid is returned to the distillation column for further separation. Therefore, the higher the reflux percentage, the greater amount of liquid is sent back to the distillation column for further separation. It follows then that the highest composition of the light component collected in the distillate is proportional to the highest possible reflux percentage. Unfortunately, at 100% reflux no distillate is collected. The figure below is an illustration of reflux. Reflux Condenser 1 Distillate collected 4 Figure 2. Diagram Illustrating Reflux.
8 An energy study on the distillation column can be simplified if an overall energy balanced is used on the distillation column. A simplified illustration of the distillation column is given in the following figure, Figure 3. Q condenser Q loss Q reboiler Figure 3. Simplified Model of the Distillation Column At steady-state, the energy added to the distillation column is equal to the energy removed from the distillation column minus any heat loss in the distillation column. The following equation was used to find the overall heat loss in the distillation column. Q reboiler = Q condenser + Q loss (2). Q reboiler is the energy added to the distillation column from the reboiler, Q condenser is the energy removed by the condenser, and Q loss is the energy loss in the distillation column.
9 m Q condenser Condensation Droplets T out m T in Vapor from Tray 1 Condensed Liquid Figure 4. Simplified Model of the Condenser Located at the Top of the Distillation Column In order to perform an energy balance on the condenser, the following equation was used: Q = mc T (3), P where Q is the energy in watts removed by the condenser, m is the mass flow rate of the entering cold water, and C p is the heat capacity of water. T is the change in temperature in degree Celsius, or more specifically, T = T out - T in (4).
10 EQUIPMENT: The distillation column is approximately 15 feet high with a diameter of approximately 6 inches. There are 12 trays within the distillation column. The section of the distillation column above the feed is known as the rectifying section. The section of the distillation column below the feed is known as the stripping section. The reboiler is located at the bottom of the column. Inside the reboiler is a heating unit composed of calrod heaters that is used to heat the mixture that is fed into the distillation column. When this mixture is heated the light component, the component of the mixture that has the lowest boiling point, vaporizes and condenses on the tray directly above the reboiler. From the heat added to the reboiler, each subsequent tray undergoes vaporization and condensation so that the top of the column has a high concentration of the lightest component and the bottom of the column has a high concentration of the heavy component (the component with the higher boiling point). Figure 4 best illustrates this separation. Liquid from tray above Vapor leaving tray Typical Tray Liquid leaving tray Vapor from tray below Figure 5. A Typical Tray. This distillation column can be run via batch operation or continuous operation. In batch distillation, a batch of chemicals, in this case methanol and water, is first fed into the column and the liquid part of this mixture settles in the reboiler. Once the reboiler is filled with the desired finite batch of the binary mixture, energy is added to the reboiler in the form of heat to start the experiment. A schematic drawing of the distillation column under batch distillation is given in Figure 6. In continuous distillation, the feed enters the column continuously. Heat is constantly supplied to the reboiler, and a continuous flow of distillate and bottoms product is collected or returned to the feed tank. A schematic drawing of the distillation column under continuous distillation is given in Figure 7.
11 Heat Removed from the Condenser Reflux Condenser 1 Distillate collected Distillate Pump Rectifying Section Feed Tank Reboiler Pump Vapor Boil-up Reboiler filled with batch of binary mixture Reboiler Heat added to the Reboiler Bottom s Product
12 Figure 6. Distillation Column Under Batch Operation Heat Removed from the Condenser Reflux Condenser 1 Distillate collected Rectifying Section Distillate Pump Feed Feed Pump Feed Tank Reboiler Pump Stripping Section Vapor Boil-up Reboiler Heat added to the Reboiler Bottom s Product
13 Figure 7. Distillation Column Under Continuous Distillation PROCEDURE: Seven experiments were ran for batch operation. The inputs required for batch distillation include Reflux Percent and Heat Added to the Reboiler. Table 1 depicts the experiments ran for batch operation of the distillation column. Table 1. Experiments for Batch Distillation Experiment Number Reflux Rate Heat Added to Reboiler, Watts Composition of batch, wt % methanol 1 100% % 2 95% % 3 95% % 4 90% % 5 70% % 6 50% % 7 20% % Four experiments were run for continuous distillation. The inputs for continuous operation include Reflux Percent, Heat added to the Reboiler, and a feed pump setting. It should be noted that a P-only controller maintained a level of approximately 11 liters in the reboiler. Table 2 depicts the experiments ran for continuous operation of the distillation column. Table 2. Experiments for Continuous Distillation Experiment Number Feed Pump Setting Reflux Rate Heat Added to Reboiler, Watts Composition of Feed, wt % methanol % % % % % % % % Distillate Flow Rate Study For batch distillation, the flow rate for the distillate was found by two different methods, manually and computer. Manual Method - The real distillate flow rate was recorded physically by manually recording the level of the distillate collected in the 1000mL beaker every 5 minutes. Computer Method The distillate flow rate given by the computer, i.e. LabVIEW is automatically recorded to a raw data sheet that is downloadable at
14 Composition of Methanol in the Distillate The composition of the distillate was found using two different methods, density and temperature data. For density calculations for batch and continuous distillation experiments, the initial distillate that was collected in the 1000mL beaker was transferred to a 50mL beaker. This distillate was used to wash a 5mLgraduated cylinder. After 3 washings, the collected distillate was poured into the 5mL-graduated cylinder to the 5mL mark. This graduated cylinder was then weighed on a triple beam balance to the nearest 100 th of a gram. Then, using the following equation, Density = Mass/volume (1) the density was found. Then, using a list of tabulated data by Perry s Handbook, 7 th edition, the composition of the distillate was found. To calculate the composition of the distillate by temperature data, the temperature of the top tray was used in conjunction with the chart given in Figure 1. The composition of the distillate was recorded as the composition of the vapor at the tray 1 of the distillation column. For batch distillation experiments, the composition of the distillate calculated from temperature data was found approximately 5 minutes after the first distillate started to flow. For continuous distillation experiments, the composition of the distillate calculated from temperature data was found at steady-state. Composition of Methanol on Each Tray Temperature data in conjunction with the chart given in Figure 1was used to find the composition of methanol in each tray. For batch distillation experiments, the composition of the distillate was found approximately 5 minutes after the first distillate started to flow. For continuous distillation experiments, the composition of the distillate calculated was found at steady-state. Energy Study on the Condenser The needed information for conducting an energy study on the condenser was Cold Water Flow Rate to the condenser, the Cold Water Supply Temperature to the condenser, and the Cold Water Return Temperature to the condenser. The following equation was used to calculate the energy removed by the condenser, Q = mc T (3), P where Q is the energy in watts removed by the condenser, m is the mass flow rate of the entering cold water, and C p is the heat capacity of water. T is the change in temperature in degree Celsius, or more specifically, T = T out - T in (4). where T in is the Cold Water Supply Temperature and T out is the Cold Water Return Temperature.
15 Overall Energy Loss in the Distillation Column At steady-state, the energy added to the distillation column is equal to the energy removed from the distillation column minus any heat loss in the distillation column. The following equation was used to find the overall heat loss in the distillation column. Q reboiler = Q condenser + Q loss (2). Q reboiler is the energy added to the distillation column from the reboiler, Q condenser is the energy removed by the condenser, and Q loss is the energy loss in the distillation column. RESULTS: The distillate flow rate was found for batch distillation manually (manually recording the distillate collected in the 1000mL beaker every 5 minutes) and then compared to the data recorded by LabVIEW (the distillate level calculated by the LabVIEW program using a pressure sensor at the bottom of the beaker containing the distillate). Table 3 and Table 4 give the results of the flow rate analysis. Table 3. Distillate Flow Rate for Batch Distillation for a Constant Reflux Rate Heat Added to Reboiler, Watts Reflux Rate LabVIEW Flow Rate, ml/min Manual Flow Rate, ml/min % % Table 4. Distillate Flow Rate for Batch Distillation for a Constant Heat Added to Reboiler Reflux Rate Heat Added to Reboiler, Watts LabVIEW Flow Rate, ml/min Manual Flow Rate, ml/min 95% %
16 Figure 8 displays the difference in flow rate of the distillate for batch distillation at a constant reflux percentage of 95%. The data displayed below represents the manually collected data. Heat added to the reboiler is the variable studied in Figure 8. As noticed in the figure below, the higher distillate flow rate is for 2500 Watts added to the reboiler. Batch Distillation Flow Rate for 2000 Watts vs. 2500Watts (95% Reflux) Watts V = 5.1t Volume of Distillate (ml) Watts V = 3.9t Time (min) Figure 8. Manual Distillate Flow Rate for Constant Reflux Rate
17 Figure 9 represents the data collected manually for batch distillation at a constant wattage supplied to the reboiler of 2500 Watts. Reflux percentage is the variable of interest. As noticed in the figure below, the higher distillate flow rate is associated with the lower reflux percentage. Batch Distillation Flow Rate of 95% vs. 20% Reflux (2500Watts) % Reflux V = 60.7t Volume of Distillate (ml) % Reflux V = 5.1t Time (min) Figure 9. Manual Distillate Flow Rate for Constant Heat Added to Reboiler
18 Figure 10 represents the comparison of the manual data and computer data for the distillate flow rate for batch distillation. The wattage supplied to the reboiler and the Reflux Percentage are held constant at 2000Watts and 95%, respectively. As noticed in the figure below, the higher distillate flow rate is associated with the manual data. Batch Distillation Manual vs. LabVIEW (2000 Watts, 95% Reflux Rate) LabVIEW V = 2.5t Volume of Distillate (ml) Manual V = 3.9t Time (min) Figure 10. Comparison of Manual and LabVIEW Distillate Flow Rate
19 Figure 11 represents the comparison of the manual data and computer data for the distillate flow rate for batch distillation. The wattage supplied to the reboiler and the Reflux Percentage are held constant at 2500Watts and 20%, respectively. As noticed in the figure below, the higher distillate flow rate is associated with the manual data. Batch Distillation Manual vs. LabVIEW (2500 Watts, 20% Reflux Rate) 2500 Manual 2000 V = 60.7t Volume of Distillate (ml) LabVIE W V = 30.8t Time (min) Figure 11. Comparison of Manual and LabVIEW Distillate Flow Rate
20 Figure 12 represents the comparison of the manual data and computer data for the distillate flow rate for batch distillation. The wattage supplied to the reboiler and the Reflux Percentage are held constant at 2500Watts and 95%, respectively. As noticed in the figure below, the higher distillate flow rate is associated with the manual data. Batch Distillation Manual vs. LabVIEW (2500 Watts, 95% Reflux Rate) LabVIEW V = 3.4t Volume of Distillate (ml) Manual V = 5.1t Time (min) Figure 12. Comparison of Manual and LabVIEW Distillate Flow Rate
21 As noticed in Figures 10 through 12, the manually collected data does not match the computer data for distillate flow rate. Assuming that the manually collected data for the distillate flow rate is the true distillate flow rate, an equation needs to be created that corrects the computer data to match the manual data. Heat Added to Reboiler, Watts Reflux Percentage Table 5. Ratio of Flow Rates for Manual and LabVIEW LabVIEW Flow Rate, ml/min Manual Flow Rate, ml/min Ratio of Flow Rates, Manual/LabVIEW Intercepts % % % Average Student T Error ±1.08 ±11.5 Relationship between Manual Flow Rate and LabVIEW Flow Rate: Manual Flow Rate = (1.68±1.08)*LabVIEW Flow 188(±11.5) Density study for methanol composition It was first believed that density would prove to be a good comparison with the temperature method for methanol composition in the distillate. However, in light of the obvious high error in density calculations, comparisons are not made between the temperature calculations for methanol composition and density calculations for methanol composition. Table 6 summarizes the error analysis for the density data at a Reflux of 50%. Reflux Percentage Table 6. Percent Methanol in Distillate from Density Calculations Volume in Graduated Cylinder (ml) Weight of Distillate (grams) Density of Distillate (grams/ml) Composition of Distillate (wt%) 50% % 50% % 50% % 50% % Students T- Error 17.1%
22 Table 7 summarizes the density calculations for batch distillation. It should be noted that these methanol compositions appear to be bogus. Table 7. Density Calculations for Methanol Composition in the Distillate for Batch Distillation Reflux Percentage Composition of Distillate (wt%) 95% 110% 90% 98% 70% 89% 50% 61% 20% 89% Table 8 summarizes the density calculations for continuous distillation. It should be noted that these methanol compositions also appear to be bogus. Table 8. Density Calculations for Methanol Composition in the Distillate for Continuous Distillation Reflux Percentage Composition of Distillate (wt%) 90% 61% 70% 57% 50% 106% Table 9 summarizes the distillate composition found for batch distillation using the temperature method. It should be noted that these calculations were conducted approximately 5 minutes after the first distillate started to flow. Table 9. Comparison of Distillate Composition for Batch Distillation with 2500 Watts added to the Reboiler Reflux Percentage Composition of Distillate, mole fraction Methanol 95% % % % % 0.18
23 Table 10 summarizes the distillate composition found for continuous distillation using the temperature method. It should be noted that these calculations were performed once the continuous distillation experiment reached steady-state. Table 10. Comparison of Distillate Composition for Continuous Distillation with 2500 Watts added to the Reboiler Reflux Percentage Composition of Distillate, mole fraction Methanol 90% % % 0.23 Table 11 presents the comparison between calculated compositions of methanol in the distillate for batch distillation and continuous distillation. Batch distillation calculations were conducted approximately 5 minutes after the first distillate started to flow. Continuous distillation calculations were conducted once steady-state was reached. Table 11. Comparison of Distillate Composition for Continuous Distillation and Batch Distillation with 2500 Watts added to the Reboiler Reflux Percentage Composition of Batch Distillate, mole fraction Methanol Composition of Continuous Distillate, mole fraction Methanol 90% % %
24 Figure 13 is graph of the methanol composition in the liquid for each tray. Figure 13 illustrates the difference in liquid composition for batch distillation with a constant reflux percentage of 95%. The variable of interest is the wattage added to the reboiler, which in this graph is 2000Watts and 2500Watts. It should be noted that these calculations were conducted approximately 5 minutes after the first distillate started to flow. Batch Distillation at 95% Reflux Liquid Composition Watts 0.8 mole fraction methanol Watts Tray Figure 13. Liquid Composition of Methanol in Each Tray for Batch Distillation
25 Figure 14 is graph of the methanol composition in the liquid for each tray. Figure 14 illustrates the difference in liquid composition for batch distillation with a constant wattage supplied to the reboiler of 2500Watts. The variable of interest is the reflux percentage. It should be noted that these calculations were conducted approximately 5 minutes after the first distillate started to flow. Batch Distillation at 2500 Watts added to Reboiler - Liquid Composition % Reflux 90% Reflux 0.7 mole fraction methanol % Reflux 95% Reflux % Reflux Tray Figure 14. Liquid Composition of Methanol in Each Tray for Batch Distillation
26 Figure 15 is graph of the methanol composition in the liquid for each tray. Figure 15 illustrates the difference in liquid composition for continuous distillation with a constant wattage supplied to the reboiler of 2500Watts. The variable of interest is the reflux percentage. It should be noted that these calculations were performed once the continuous distillation experiment reached steady-state. Continuous Distillation at 2500 Watts added to Reboiler - Liquid Composition % Reflux mole fraction methanol % Reflux 70% Reflux % Reflux Tray Figure 15. Liquid Composition of Methanol in Each Tray for Continuous Distillation
27 Energy Balance on the Condenser and Heat Loss in the Distillation Column It should be noted that during the course of this study, it was found that the cold water flow rate displayed by the computer is incorrect. Table 12 summarizes theses results Table 12. Cold Water Flow Rate Study Cold Water Flow Rate Displayed Cold Water Flow Rate found Ratio of Manual/LabVIEW by LabVIEW (L/min) Manually (L/min) Therefore, it follows that the correction factor for the flow rate given by the computer is 2.8 Table 13 is a summary of the energy study for batch distillation experiments. Table 13. Summary of Energy Study for Batch Distillation Reflux Rate Heat Added to Reboiler, Watts Heat Removed by Condenser, Watts Heat Loss in Column, Watts 100% % % % % % Table 13 is a summary of the energy study for the continuous distillation experiments Table 13. Summary of Energy Study for Continuous Distillation Reflux Rate Heat Added to Reboiler, Watts Heat Removed by Condenser, Watts Heat Loss in Column, Watts 95% % % %
28 Figure 16 is a graph comparing the heat absorbed by the condenser for batch distillation experiments and continuous distillation experiments. The wattage supplied to the reboiler is 2500 Watts for all data graphed below. Heat absorbed by Condenser Vs. Reflux % Heat absorbed by condenser (Watts) Batch Continuous Reflux % Figure 16. Comparison of Heat Absorbed by Condenser for Batch and Continuous Distillation
29 DISCUSSION OF RESULTS: When comparing the distillate flow rates for the Manual and LabVIEW methods, the distillate flow rate displayed by the computer, i.e. LabVIEW, is consistently lower than the real distillate flow rate, i.e. Manually. Specifically, the real distillate flow rate is 1.68±1.08 times the computer distillate flow rate. The correction for this discrepancy was found to be Q corrected = (1.68±1.08)Q LabVIEW - 188(±11.5), where Q is the flow rate. When comparing the distillate flow rates for batch distillation at different reflux percentages but at a constant wattage supplied to the reboiler, the higher distillate flow rate is due to the lower reflux percentage, as expected in the theory. When comparing the distillate flow rates for batch distillation at different wattages added to the reboiler but at a constant reflux percent, the higher distillate flow rate is due to a higher wattage supplied to the reboiler. The composition of the methanol in the distillate was found using two different methods, density and temperature. The compositions found using the density calculations proved to be bogus with a Student-T error of 17.4% for batch distillation with 50% reflux and 2500Watts added to the reboiler. Commonly the composition of the methanol exceeded 100 wt%. Obviously, it is impossible to have greater than 100 wt% methanol in the distillate. Also, the composition of methanol in the distillate would inconsistently increase or decrease as the reflux percentage decreased. Therefore, the density calculations proved erroneous. The composition of methanol in the distillate found from temperature calculations proved to be consistent with the theory. An increase in percent methanol is related to an increase in reflux percentage, and a decrease in percent methanol in the distillate is related to a decrease in reflux percentage. Batch distillation experiments give a higher composition of methanol in the distillate than continuous distillation experiments when reflux percentage and heat added to the reboiler are equal. It should be noted that batch distillation compositions occurred approximately 5 minutes after the first distillate started to flow and the continuous distillation compositions occurred at steady-state. The composition of methanol in each tray found from temperature calculations also proved to be consistent with the theory. Tray 1 contained the highest percent methanol and each subsequent tray contained successively less methanol. There is little difference in corresponding tray compositions for reflux percentages of 90% and 95% for batch distillation. The same phenomenon was also observed for continuous distillation experiments at 90% and 95% reflux. When comparing the methanol composition on each tray for batch distillation at different wattages added to the reboiler but at a constant reflux percent, the methanol concentration in the lower trays (7 through 12) are lower for the higher wattage supplied to the reboiler. This is due to the fact that there is a greater flow rate of vapor to the top of the column with a higher wattage supplied to the reboiler. For batch distillation experiments, the energy removed by the condenser is greater than the energy removed from the condenser during continuous distillation experiments.
30 CONCLUSIONS: The distillate flow rate found by LabVIEW, i.e. the computer is incorrect and does not match the manually calculated flow rate, which is accepted as the real distillate flow rate. The correction for this discrepancy was found to be Q corrected = (1.68±1.08)Q LabVIEW - 188(±11.5), where Q is the flow rate is L/min. Methanol concentration increases in the trays as it ascends in the distillation column. Batch distillation experiments give a higher initial methanol composition in the distillate than continuous distillation experiments. At lower reflux percentages, the distillate flow rate is higher for both continuous distillation experiments and batch distillation experiments. Also, at lower reflux percentages, the concentration of methanol is less in the stripping section (trays 7 through 12) of the distillation column for both batch and continuous distillation experiments. At higher watts added to the reboiler, there is a corresponding lower concentration of methanol in the lower trays (trays 7 through 12) of the distillation column for batch distillation experiments. RECOMMENDAONS: It would be of interest to observe continuous distillation at a constant reflux percentage but varying the wattage supplied to the reboiler. Also, it would be of interest to observe continuous distillation for different feed pump settings. In addition, a study should be conducted to determine why the heat absorbed by the condenser is less for continuous distillation than for batch distillation. REFERENCES: Unit Operations of Chemical Engineering, 6 th Ed., Warren L. McCabe, Julian C. Smith and Peter Harriott, McGraw-Hill, Boston, Perry, Robert H. and Don W. Green. Perry s Chemical Engineers Handbook, 7 th Edition, June 1, McGraw-Hill Professional Cunningham, James R. Dr. Lecture Notes Fall 2002 for ENCH 432 at the University of Tennessee at Chattanooga. UTC Engineering Controls Lab Online. University of Tennessee at Chattanooga. Appendices
31 Batch Distillation at 95% Reflux Vapor Composition Watts Tray Batch Distillation at 2500 Watts added to Reboiler - Vapor Composition % Reflux 100% 95% % 90% % Reflux Tray
32 Continuous Distillation at 2500 Watts added to Reboiler - Vapor Composition % % 90% % Tray
Fractional Distillation Lab Simulating The Refining of Petroleum 12/12 Integrated Science 3 Redwood High School Name : Per:
 Simulating The Refining of Petroleum 12/12 Integrated Science 3 Redwood High School Name : Per: Introduction Petroleum, or crude oil, is a complex mixture of substances. It is believed that crude oil is
Simulating The Refining of Petroleum 12/12 Integrated Science 3 Redwood High School Name : Per: Introduction Petroleum, or crude oil, is a complex mixture of substances. It is believed that crude oil is
Roger Adams Chemical 600 S. Mathews Urbana, IL REF: Methanol Recovery via Distillation Rotation 1 Assignment Memo
 Roger Adams Chemical 600 S. Mathews Urbana, IL 61801 DATE: 06-October-2005 TO: Marina Miletic, Head Engineering Manager Scott Slifka, Lab Supervisor FROM: Vincent Ho, Process Engineer Mark Butzow, Process
Roger Adams Chemical 600 S. Mathews Urbana, IL 61801 DATE: 06-October-2005 TO: Marina Miletic, Head Engineering Manager Scott Slifka, Lab Supervisor FROM: Vincent Ho, Process Engineer Mark Butzow, Process
Methanol distribution in amine systems and its impact on plant performance Abstract: Methanol in gas treating Methanol impact on downstream units
 Abstract: Presented at the AIChE Spring 2015 meeting in Austin, TX, USA Methanol distribution in amine systems and its impact on plant performance Anand Govindarajan*, Nathan A. Hatcher, and Ralph H. Weiland
Abstract: Presented at the AIChE Spring 2015 meeting in Austin, TX, USA Methanol distribution in amine systems and its impact on plant performance Anand Govindarajan*, Nathan A. Hatcher, and Ralph H. Weiland
Synthesis of Optimal Batch Distillation Sequences
 Presented at the World Batch Forum North American Conference Woodcliff Lake, NJ April 7-10, 2002 107 S. Southgate Drive Chandler, Arizona 85226-3222 480-893-8803 Fax 480-893-7775 E-mail: info@wbf.org www.wbf.org
Presented at the World Batch Forum North American Conference Woodcliff Lake, NJ April 7-10, 2002 107 S. Southgate Drive Chandler, Arizona 85226-3222 480-893-8803 Fax 480-893-7775 E-mail: info@wbf.org www.wbf.org
MONITORING AND RESEARCH DEPARTMENT
 MONITORING AND RESEARCH DEPARTMENT REPORT NO. 10-01 EVALUATION OF THE SETTLING CHARACTERISTICS OF NORTH SIDE WATER RECLAMATION PLANT COMBINED SOLIDS AND STICKNEY WATER RECLAMATION PLANT PRELIMINARY SLUDGE
MONITORING AND RESEARCH DEPARTMENT REPORT NO. 10-01 EVALUATION OF THE SETTLING CHARACTERISTICS OF NORTH SIDE WATER RECLAMATION PLANT COMBINED SOLIDS AND STICKNEY WATER RECLAMATION PLANT PRELIMINARY SLUDGE
Production of Dimethyl Ether
 Production of Dimethyl Ether Background A feasibility study on the production of 99.5 wt% dimethyl ether (DME) is to be performed. The plant is capable of producing 50,000 metric tons of DME per year via
Production of Dimethyl Ether Background A feasibility study on the production of 99.5 wt% dimethyl ether (DME) is to be performed. The plant is capable of producing 50,000 metric tons of DME per year via
Estimation Procedure for Following Vapor Pressure Changes
 Estimation Procedure for Following Vapor Pressure Changes through Repeated Blending of Petroleum Stocks from Boiling Point Curves A practical method to estimate the vapor pressures of blended and reblended
Estimation Procedure for Following Vapor Pressure Changes through Repeated Blending of Petroleum Stocks from Boiling Point Curves A practical method to estimate the vapor pressures of blended and reblended
Biodiesel Production and Analysis
 Biodiesel Production and Analysis Introduction A key current focus in science and engineering is the development of technologies for generating and utilizing new sources of energy. Climate change, geopolitics,
Biodiesel Production and Analysis Introduction A key current focus in science and engineering is the development of technologies for generating and utilizing new sources of energy. Climate change, geopolitics,
Biodiesel. As fossil fuels become increasingly expensive to extract and produce, bio-diesel is
 Aaron Paternoster CHEM 380 10D Prof. Laurie Grove January 30, 2015 Biodiesel Introduction As fossil fuels become increasingly expensive to extract and produce, bio-diesel is proving to be an economically
Aaron Paternoster CHEM 380 10D Prof. Laurie Grove January 30, 2015 Biodiesel Introduction As fossil fuels become increasingly expensive to extract and produce, bio-diesel is proving to be an economically
As the global energy sector
 Improved distillation efficiency Dividing wall technology applied to a xylenes separation project delivered superior energy efficiency compared to a two- arrangement MANISH BHARGAVA, ROOMI KALITA and JOSEPH
Improved distillation efficiency Dividing wall technology applied to a xylenes separation project delivered superior energy efficiency compared to a two- arrangement MANISH BHARGAVA, ROOMI KALITA and JOSEPH
White Paper.
 The Advantage of Real Atmospheric Distillation Complying with the ASTM D7345 Test Method in the Distillation Process Introduction / Background In the past, refiners enjoyed a constant supply of the same
The Advantage of Real Atmospheric Distillation Complying with the ASTM D7345 Test Method in the Distillation Process Introduction / Background In the past, refiners enjoyed a constant supply of the same
Production of Biodiesel from Waste Oil via Catalytic Distillation
 Production of Biodiesel from Waste Oil via Catalytic Distillation Zhiwen Qi, Yuanqing Liu, Blaise Pinaud, Peter Rehbein Flora T.T. Ng*, Garry L. Rempel Department of Chemical Engineering, University of
Production of Biodiesel from Waste Oil via Catalytic Distillation Zhiwen Qi, Yuanqing Liu, Blaise Pinaud, Peter Rehbein Flora T.T. Ng*, Garry L. Rempel Department of Chemical Engineering, University of
Atmospheric Crude Tower with Aspen HYSYS V8.0
 Atmospheric Crude Tower with Aspen HYSYS V8.0 1. Lesson Objectives Assign petroleum assay to stream Configure column pre-heater Configure crude tower 2. Prerequisites Aspen HYSYS V8.0 Introduction to distillation
Atmospheric Crude Tower with Aspen HYSYS V8.0 1. Lesson Objectives Assign petroleum assay to stream Configure column pre-heater Configure crude tower 2. Prerequisites Aspen HYSYS V8.0 Introduction to distillation
Improving the Quality and Production of Biogas from Swine Manure and Jatropha (Jatropha curcas) Seeds
 Improving the Quality and Production of Biogas from Swine Manure and Jatropha (Jatropha curcas) Seeds Amy Lizbeth J. Rico Company: Tarlac Agricultural University College of Engineering Technology Address:
Improving the Quality and Production of Biogas from Swine Manure and Jatropha (Jatropha curcas) Seeds Amy Lizbeth J. Rico Company: Tarlac Agricultural University College of Engineering Technology Address:
Heating Methods. Reflux and Distillation
 Heating Methods Reflux and Distillation Heating Methods Reflux Distillation Reflux You will use this next lab for the synthesis of aspirin not in this lab experiment Heating the reaction contents without
Heating Methods Reflux and Distillation Heating Methods Reflux Distillation Reflux You will use this next lab for the synthesis of aspirin not in this lab experiment Heating the reaction contents without
Optimization of Toluene-Benzene Reactor
 Optimization of Toluene-Benzene Reactor Aaron Vigil & Eric Vasko 10/28/2013 1 Introduction The production of benzene is an important process for the petrochemical industry. This chemical process will use
Optimization of Toluene-Benzene Reactor Aaron Vigil & Eric Vasko 10/28/2013 1 Introduction The production of benzene is an important process for the petrochemical industry. This chemical process will use
Evaluation of phase separator number in hydrodesulfurization (HDS) unit
 IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS Evaluation of phase separator number in hydrodesulfurization (HDS) unit To cite this article: A D Jayanti and A Indarto 2016 IOP
IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS Evaluation of phase separator number in hydrodesulfurization (HDS) unit To cite this article: A D Jayanti and A Indarto 2016 IOP
BTX Extractive Distillation Capacity Increased by Enhanced Packing Distributors
 BTX Extractive Distillation Capacity Increased by Enhanced Packing Distributors Karl Kolmetz kkolmetz@yahoo.com Jeff Gray jeffngray@hotmail.com Mel Chua Sulzer Chemtech Raghu Desai Sulzer Chemtech AIChE
BTX Extractive Distillation Capacity Increased by Enhanced Packing Distributors Karl Kolmetz kkolmetz@yahoo.com Jeff Gray jeffngray@hotmail.com Mel Chua Sulzer Chemtech Raghu Desai Sulzer Chemtech AIChE
SYNTHESIS OF BIODIESEL
 SYNTHESIS OF BIODIESEL AIM 1. To generate laboratory know-how for the process of production of biodiesel from the given oil feed stock 2. To perform basic mass and energy balance calculations for a large
SYNTHESIS OF BIODIESEL AIM 1. To generate laboratory know-how for the process of production of biodiesel from the given oil feed stock 2. To perform basic mass and energy balance calculations for a large
HEAT PUMPS Advanced Distillation - GT-HIDS (Heat Integrated Differential Separation)
 HEAT PUMPS Advanced Distillation - GT-HIDS (Heat Integrated Differential Separation) Joseph C. Gentry, GTC Technology Vice President Technology & Engineering ERTC - Lisbon 15 November 2016 What is HIDS?
HEAT PUMPS Advanced Distillation - GT-HIDS (Heat Integrated Differential Separation) Joseph C. Gentry, GTC Technology Vice President Technology & Engineering ERTC - Lisbon 15 November 2016 What is HIDS?
Dimethyl Ether Production Material Balances
 Dimethyl Ether Production Material Balances Dimethyl ether (DME) is used primarily as a propellant. It is miscible with most organic solvents and has high solubility with water. Recently, the use of DME
Dimethyl Ether Production Material Balances Dimethyl ether (DME) is used primarily as a propellant. It is miscible with most organic solvents and has high solubility with water. Recently, the use of DME
Biodiesel: Making Renewable Fuel from Waste Oils
 Biodiesel: Making Renewable Fuel from Waste Oils Author/School: Matt Steiman, Wilson College, Chambersburg PA Introduction Biodiesel is a renewable fuel made from any biologically based oil, and can be
Biodiesel: Making Renewable Fuel from Waste Oils Author/School: Matt Steiman, Wilson College, Chambersburg PA Introduction Biodiesel is a renewable fuel made from any biologically based oil, and can be
TKP4105/TKP4110 Oldershow perforated plate distillation Report
 TKP4105/TKP4110 Oldershow perforated plate distillation Report Audun F. Buene audunfor@stud.ntnu.no Elise Landsem elisel@stud.ntnu.no Group B19 Supervisor: Vladmiros Minasidis Laboratory: K4-213 Conducted
TKP4105/TKP4110 Oldershow perforated plate distillation Report Audun F. Buene audunfor@stud.ntnu.no Elise Landsem elisel@stud.ntnu.no Group B19 Supervisor: Vladmiros Minasidis Laboratory: K4-213 Conducted
Performance of Biodiesel Fuel in cold weather condition. Mechanical Engineering Graduation Thesis / Presented by: Zeloon Lye
 Performance of Biodiesel Fuel in cold weather condition Mechanical Engineering Graduation Thesis 25.416/25.475 Presented by: Zeloon Lye 6805215 1 Introduction of biodiesel fuel Biodiesel is a clean burning
Performance of Biodiesel Fuel in cold weather condition Mechanical Engineering Graduation Thesis 25.416/25.475 Presented by: Zeloon Lye 6805215 1 Introduction of biodiesel fuel Biodiesel is a clean burning
Biodiesel Production and Analysis
 Biodiesel Production and Analysis Introduction A key current focus in science and engineering is the development of technologies for generating and utilizing new sources of energy. Climate change, geopolitics,
Biodiesel Production and Analysis Introduction A key current focus in science and engineering is the development of technologies for generating and utilizing new sources of energy. Climate change, geopolitics,
Oregon DOT Slow-Speed Weigh-in-Motion (SWIM) Project: Analysis of Initial Weight Data
 Portland State University PDXScholar Center for Urban Studies Publications and Reports Center for Urban Studies 7-1997 Oregon DOT Slow-Speed Weigh-in-Motion (SWIM) Project: Analysis of Initial Weight Data
Portland State University PDXScholar Center for Urban Studies Publications and Reports Center for Urban Studies 7-1997 Oregon DOT Slow-Speed Weigh-in-Motion (SWIM) Project: Analysis of Initial Weight Data
IC18DV/92/SCP - Multipurpose Distillation Plant with Automatic Control
 CHEMICAL ENGINEERING IC18DV/92/SCP - Multipurpose Distillation Plant with Automatic Control - Code 991215 IC18DV/92/SCP - Multipurpose Distillation Plant with Automatic Control DIDACTA Italia S.r.l. -
CHEMICAL ENGINEERING IC18DV/92/SCP - Multipurpose Distillation Plant with Automatic Control - Code 991215 IC18DV/92/SCP - Multipurpose Distillation Plant with Automatic Control DIDACTA Italia S.r.l. -
ProSimTechs PROCESS SIMULATION TECHNICS
 PROCESS SIMULATION TECHNICS Rigorous Process Simulation Services for the Vegetable oils & Fats refining industry (Degumming - Neutralising Bleaching Deodorising/Physical Refining) Deodorising and Physical
PROCESS SIMULATION TECHNICS Rigorous Process Simulation Services for the Vegetable oils & Fats refining industry (Degumming - Neutralising Bleaching Deodorising/Physical Refining) Deodorising and Physical
INVESTIGATION ON VISBREAKING-RESIDUE AND FINISHED FUEL OIL PRODUCT CLOSED CUP FLASH POINT
 Petroleum & Coal ISSN 1337-7027 Available online at www.vurup.sk/pc Petroleum & Coal 51 (4) 277-281, 2009 INVESTIGATION ON VISBREAKING-RESIDUE AND FINISHED FUEL OIL PRODUCT CLOSED CUP FLASH POINT Dicho
Petroleum & Coal ISSN 1337-7027 Available online at www.vurup.sk/pc Petroleum & Coal 51 (4) 277-281, 2009 INVESTIGATION ON VISBREAKING-RESIDUE AND FINISHED FUEL OIL PRODUCT CLOSED CUP FLASH POINT Dicho
CHEMISTRY 135. Biodiesel Production and Analysis
 CHEMISTRY 135 General Chemistry II Biodiesel Production and Analysis The energy content of biodiesel can be roughly estimated with a simple laboratory apparatus. What features of biodiesel make it an attractive
CHEMISTRY 135 General Chemistry II Biodiesel Production and Analysis The energy content of biodiesel can be roughly estimated with a simple laboratory apparatus. What features of biodiesel make it an attractive
Storage, Bulk Transfer, and In-Plant Handling of Zeotropic Refrigerant Blends
 Purdue University Purdue epubs International Refrigeration and Air Conditioning Conference School of Mechanical Engineering 1996 Storage, Bulk Transfer, and InPlant Handling of Zeotropic Refrigerant Blends
Purdue University Purdue epubs International Refrigeration and Air Conditioning Conference School of Mechanical Engineering 1996 Storage, Bulk Transfer, and InPlant Handling of Zeotropic Refrigerant Blends
International Journal of Advanced Engineering Technology E-ISSN
 International Journal of Advanced Engineering Technology E-ISS 976-3945 Research Article EXPERIMETAL WORKSTUDY O THE EFFECT OF ETHAOL GASOLIE BLEDS O THE PERFORMACE OF TWO STROKE PETROL EGIE Prof. Viral
International Journal of Advanced Engineering Technology E-ISS 976-3945 Research Article EXPERIMETAL WORKSTUDY O THE EFFECT OF ETHAOL GASOLIE BLEDS O THE PERFORMACE OF TWO STROKE PETROL EGIE Prof. Viral
CENTRIFUGAL PUMP: Parallel and Series Operation 11/11/02
 CENTRIFUGAL PUMP: Parallel and Series Operation 11/11/02 1 CENTRIFUGAL PUMP Location Sub-basement SB-92. (Manual is available) Introduction This experiment illustrates the basic operation and characteristics
CENTRIFUGAL PUMP: Parallel and Series Operation 11/11/02 1 CENTRIFUGAL PUMP Location Sub-basement SB-92. (Manual is available) Introduction This experiment illustrates the basic operation and characteristics
Experimental Investigation and Modeling of Liquid-Liquid Equilibria in Biodiesel + Glycerol + Methanol
 11 2nd International Conference on Chemical Engineering and Applications IPCBEE vol. 23 (11) (11) IACSIT Press, Singapore Experimental Investigation and Modeling of Liquid-Liquid Equilibria in + + Methanol
11 2nd International Conference on Chemical Engineering and Applications IPCBEE vol. 23 (11) (11) IACSIT Press, Singapore Experimental Investigation and Modeling of Liquid-Liquid Equilibria in + + Methanol
Microchannel Distillation of JP-8 Jet Fuel for Sulfur Content Reduction
 Microchannel Distillation of JP-8 Jet Fuel for ulfur Content Reduction Feng Zheng, Victoria. tenkamp, Ward E. TeGrotenhuis, Xiwen Huang and David L. King, Pacific Northwest National Laboratory, Richland,
Microchannel Distillation of JP-8 Jet Fuel for ulfur Content Reduction Feng Zheng, Victoria. tenkamp, Ward E. TeGrotenhuis, Xiwen Huang and David L. King, Pacific Northwest National Laboratory, Richland,
METHANOL RECOVERY: BASICS, FINE TUNING, AND ENERGY BALANCE ISSUES. Dickinson College Biodiesel Shop Carlisle, PA
 METHANOL RECOVERY: BASICS, FINE TUNING, AND ENERGY BALANCE ISSUES Dickinson College Biodiesel Shop Carlisle, PA What is methanol recovery? Biodiesel and Glycerin contain surplus methanol Methanol can be
METHANOL RECOVERY: BASICS, FINE TUNING, AND ENERGY BALANCE ISSUES Dickinson College Biodiesel Shop Carlisle, PA What is methanol recovery? Biodiesel and Glycerin contain surplus methanol Methanol can be
Efficiency Increment on 0.35 mm and 0.50 mm Thicknesses of Non-oriented Steel Sheets for 0.5 Hp Induction Motor
 International Journal of Materials Engineering 2012, 2(2): 1-5 DOI: 10.5923/j.ijme.20120202.01 Efficiency Increment on 0.35 mm and 0.50 mm Thicknesses of Non-oriented Steel Sheets for 0.5 Hp Induction
International Journal of Materials Engineering 2012, 2(2): 1-5 DOI: 10.5923/j.ijme.20120202.01 Efficiency Increment on 0.35 mm and 0.50 mm Thicknesses of Non-oriented Steel Sheets for 0.5 Hp Induction
Biodiesel Fundamentals for High School Chemistry Classes. Laboratory 7: Using Differences in Solubility to Remove Contaminants from Biodiesel
 Laboratory 7: Using Differences in Solubility to Remove Contaminants from Biodiesel Topics Covered Solubility Polarity Like dissolves like Partition Ratio Equipment Needed (per pair or group) One graduated
Laboratory 7: Using Differences in Solubility to Remove Contaminants from Biodiesel Topics Covered Solubility Polarity Like dissolves like Partition Ratio Equipment Needed (per pair or group) One graduated
Crude Assay, ASTM, TBP distillations, Evaluation of crude oil properties.
 Crude Assay, ASTM, TBP distillations, Evaluation of crude oil properties. Crude Oil Distillation Crude oil distillation is an open art technology. The crude oil is distilled at atmospheric pressure and
Crude Assay, ASTM, TBP distillations, Evaluation of crude oil properties. Crude Oil Distillation Crude oil distillation is an open art technology. The crude oil is distilled at atmospheric pressure and
Aspen Hysys based Simulation and Analysis of Crude Distillation Unit
 International Journal of Current Engineering and Technology E-ISSN 2277 4106, P-ISSN 2347 5161 2015INPRESSCO, All Rights Reserved Available at http://inpressco.com/category/ijcet Research Article Shankar
International Journal of Current Engineering and Technology E-ISSN 2277 4106, P-ISSN 2347 5161 2015INPRESSCO, All Rights Reserved Available at http://inpressco.com/category/ijcet Research Article Shankar
BIODIESEL. Lesson 4d Test biodiesel in a diesel generator. Developed by Engineering and Technology Curriculum Team
 BIODIESEL Lesson 4d Test biodiesel in a diesel generator In a laboratory setting, test biodiesel in a diesel generator by estimating Btu. define energy density, kwh, and Btu calculate energy density, kwh
BIODIESEL Lesson 4d Test biodiesel in a diesel generator In a laboratory setting, test biodiesel in a diesel generator by estimating Btu. define energy density, kwh, and Btu calculate energy density, kwh
Free Pre-Algebra Lesson 44! page 1. A bottle of salad dressing, consisting of oil and vinegar.
 Free Pre-Algebra Lesson 44 page 1 Lesson 44 Percents in Mixtures Chemists may specify the strength of a solution by using a percent. For example, you can buy isopropyl alcohol at the drug store in a 91%
Free Pre-Algebra Lesson 44 page 1 Lesson 44 Percents in Mixtures Chemists may specify the strength of a solution by using a percent. For example, you can buy isopropyl alcohol at the drug store in a 91%
FEATURE ARTICLE. Advanced Function Analyzers: Real-time Measurement of Particulate Matter Using Flame Ionization Detectors. Hirokazu Fukushima
 FEATURE ARTICLE FEATURE ARTICLE Advanced Function Analyzers: Real-time Measurement of Particulate Matter Using Flame Ionization Detectors Advanced Function Analyzers: Real-time Measurement of Particulate
FEATURE ARTICLE FEATURE ARTICLE Advanced Function Analyzers: Real-time Measurement of Particulate Matter Using Flame Ionization Detectors Advanced Function Analyzers: Real-time Measurement of Particulate
Correlation of True Boiling Point of Crude Oil
 International Refereed Journal of Engineering and Science (IRJES) ISSN (Online) 2319-183X, (Print) 2319-1821 Volume 6, Issue 7 (July 2017), PP.45-49 Correlation of True Boiling Point of Crude Oil Justus
International Refereed Journal of Engineering and Science (IRJES) ISSN (Online) 2319-183X, (Print) 2319-1821 Volume 6, Issue 7 (July 2017), PP.45-49 Correlation of True Boiling Point of Crude Oil Justus
Detection of Volatile Organic Compounds in Gasoline and Diesel Using the znose Edward J. Staples, Electronic Sensor Technology
 Detection of Volatile Organic Compounds in Gasoline and Diesel Using the znose Edward J. Staples, Electronic Sensor Technology Electronic Noses An electronic nose produces a recognizable response based
Detection of Volatile Organic Compounds in Gasoline and Diesel Using the znose Edward J. Staples, Electronic Sensor Technology Electronic Noses An electronic nose produces a recognizable response based
COMPARISON OF TOTAL ENERGY CONSUMPTION NECESSARY FOR SUBCRITICAL AND SUBCRITICAL SYNTHESIS OF BIODIESEL. S. Glisic 1, 2*, D.
 COMPARISON OF TOTAL ENERGY CONSUMPTION NECESSARY FOR SUBCRITICAL AND SUBCRITICAL SYNTHESIS OF BIODIESEL S. Glisic 1, 2*, D. Skala 1, 2 1 Faculty of Technology and Metallurgy, University of Belgrade, Karnegijeva
COMPARISON OF TOTAL ENERGY CONSUMPTION NECESSARY FOR SUBCRITICAL AND SUBCRITICAL SYNTHESIS OF BIODIESEL S. Glisic 1, 2*, D. Skala 1, 2 1 Faculty of Technology and Metallurgy, University of Belgrade, Karnegijeva
Acrolein from propylene and oxygen from air [ ]
![Acrolein from propylene and oxygen from air [ ] Acrolein from propylene and oxygen from air [ ]](/thumbs/87/96044310.jpg) Acrolein from propylene and oxygen from air [107-02-8] CONTENTS OF FACTORY GATE TO FACTORY GATE LIFE CYCLE INVENTORY SUMMARY Chemistry... 2 Process Summary... 3 Summary of LCI Information... 5 Process
Acrolein from propylene and oxygen from air [107-02-8] CONTENTS OF FACTORY GATE TO FACTORY GATE LIFE CYCLE INVENTORY SUMMARY Chemistry... 2 Process Summary... 3 Summary of LCI Information... 5 Process
Learning Equipment for the Flammability Limits of Liquefied Petroleum Gas
 American Journal of Applied Sciences 9 (8): 1316-1320, 2012 ISSN 1546-9239 2012 Science Publications Learning Equipment for the Flammability Limits of Liquefied Petroleum Gas 1 Siriratchanee Sirisawat
American Journal of Applied Sciences 9 (8): 1316-1320, 2012 ISSN 1546-9239 2012 Science Publications Learning Equipment for the Flammability Limits of Liquefied Petroleum Gas 1 Siriratchanee Sirisawat
Live Crude Oil Volatility
 Live Crude Oil Volatility Dan Wispinski : Alberta Innovates Technology Futures Bob Falkiner : Imperial Oil Engineering Services CCQTA/COQA October 31, 2014 Food-Agriculture Environment Health Pipeline
Live Crude Oil Volatility Dan Wispinski : Alberta Innovates Technology Futures Bob Falkiner : Imperial Oil Engineering Services CCQTA/COQA October 31, 2014 Food-Agriculture Environment Health Pipeline
Project Reference No.: 40S_B_MTECH_007
 PRODUCTION OF BIODIESEL FROM DAIRY WASH WATER SCUM THROUGH HETEROGENEOUS CATALYST AND PERFORMANCE EVALUATION OF TBC DIESEL ENGINE FOR DIFFERENT DIESEL AND METHANOL BLEND RATIOS Project Reference No.: 40S_B_MTECH_007
PRODUCTION OF BIODIESEL FROM DAIRY WASH WATER SCUM THROUGH HETEROGENEOUS CATALYST AND PERFORMANCE EVALUATION OF TBC DIESEL ENGINE FOR DIFFERENT DIESEL AND METHANOL BLEND RATIOS Project Reference No.: 40S_B_MTECH_007
Optimal Model-Based Production Planning for Refinery Operation
 Optimal Model-Based Production Planning for Refinery Operation Abdulrahman Alattas Ignacio Grossmann Chemical Engineering Department Carnegie Mellon University EWO Meeting March 2008 1 Introduction Refinery
Optimal Model-Based Production Planning for Refinery Operation Abdulrahman Alattas Ignacio Grossmann Chemical Engineering Department Carnegie Mellon University EWO Meeting March 2008 1 Introduction Refinery
Determination of Volume Correction Factors for FAME and FAME / Mineral-diesel blends
 H & D Fitzgerald Ltd. Determination of Volume Correction Factors for FAME and FAME / Mineral-diesel blends Carried out for the Energy Institute by H&D Fitzgerald Ltd. Cefn Du Tremeirchion St. Asaph LL17
H & D Fitzgerald Ltd. Determination of Volume Correction Factors for FAME and FAME / Mineral-diesel blends Carried out for the Energy Institute by H&D Fitzgerald Ltd. Cefn Du Tremeirchion St. Asaph LL17
Live Crude Oil Volatility
 Live Crude Oil Volatility Dan Wispinski : Alberta Innovates Technology Futures Bob Falkiner : Imperial Oil Engineering Services October 16/15 PerkinElmer Corpus Christi Any and all implied or statutory
Live Crude Oil Volatility Dan Wispinski : Alberta Innovates Technology Futures Bob Falkiner : Imperial Oil Engineering Services October 16/15 PerkinElmer Corpus Christi Any and all implied or statutory
On-Line Process Analyzers: Potential Uses and Applications
 On-Line Process Analyzers: Potential Uses and Applications INTRODUCTION The purpose of this report is to provide ideas for application of Precision Scientific process analyzers in petroleum refineries.
On-Line Process Analyzers: Potential Uses and Applications INTRODUCTION The purpose of this report is to provide ideas for application of Precision Scientific process analyzers in petroleum refineries.
PEP Review METHYL TERTIARY BUTYL ETHER PRODUCTION FROM STEAM CRACKER C 4 STREAM By Syed N. Naqvi (December 2012)
 PEP Review 2012-07 METHYL TERTIARY BUTYL ETHER PRODUCTION FROM STEAM CRACKER C 4 STREAM By Syed N. Naqvi (December 2012) ABSTRACT This Review presents a technoeconomic evaluation of a methyl tertiary butyl
PEP Review 2012-07 METHYL TERTIARY BUTYL ETHER PRODUCTION FROM STEAM CRACKER C 4 STREAM By Syed N. Naqvi (December 2012) ABSTRACT This Review presents a technoeconomic evaluation of a methyl tertiary butyl
Kolmetz Handbook of Process Equipment Design REFINERY ATMOSPHERIC CRUDE TOWER SELECTION AND SIZING (ENGINEERING DESIGN GUIDELINE)
 Page: 1 of 164 SOLUTIONS, STANDARDS AND SOFTWARE www.klmtechgroup.com Rev 01 KLM Technology #03-12 Block Aronia, Jalan Sri Perkasa 2 Taman Tampoi Utama 81200 Johor Bahru Malaysia Kolmetz Handbook of TOWER
Page: 1 of 164 SOLUTIONS, STANDARDS AND SOFTWARE www.klmtechgroup.com Rev 01 KLM Technology #03-12 Block Aronia, Jalan Sri Perkasa 2 Taman Tampoi Utama 81200 Johor Bahru Malaysia Kolmetz Handbook of TOWER
Energy costs are the largest
 Improving the distillation energy network Energy-efficient design applied to the refit of a distillation unit was achieved through optimisation between the distillation column and heat network system SOUN
Improving the distillation energy network Energy-efficient design applied to the refit of a distillation unit was achieved through optimisation between the distillation column and heat network system SOUN
THE STUDY ON EFFECT OF TORQUE ON PISTON LATERAL MOTION
 THE STUDY ON EFFECT OF TORQUE ON PISTON LATERAL MOTION Vinay V. Kuppast 1, S. N. Kurbet 2, A. M. Yadawad 3, G. K. Patil 4 1 Associate Professor, 2 Professor & Head, 4 Associate Professor, Department of
THE STUDY ON EFFECT OF TORQUE ON PISTON LATERAL MOTION Vinay V. Kuppast 1, S. N. Kurbet 2, A. M. Yadawad 3, G. K. Patil 4 1 Associate Professor, 2 Professor & Head, 4 Associate Professor, Department of
Single Cylinder 4 Stroke VCR Diesel Engine Performance And Analysis At Various Blends Of Fuels Under Various Cooling Rates
 ISSN: 2278 0211 (Online) Single Cylinder 4 Stroke VCR Diesel Engine Performance And Analysis At Various Blends Of Fuels Under Various Cooling Rates B Lakshmana Swamy Associate Professor, Mechanical Engineering
ISSN: 2278 0211 (Online) Single Cylinder 4 Stroke VCR Diesel Engine Performance And Analysis At Various Blends Of Fuels Under Various Cooling Rates B Lakshmana Swamy Associate Professor, Mechanical Engineering
Thermal Stress Analysis of Diesel Engine Piston
 International Conference on Challenges and Opportunities in Mechanical Engineering, Industrial Engineering and Management Studies 576 Thermal Stress Analysis of Diesel Engine Piston B.R. Ramesh and Kishan
International Conference on Challenges and Opportunities in Mechanical Engineering, Industrial Engineering and Management Studies 576 Thermal Stress Analysis of Diesel Engine Piston B.R. Ramesh and Kishan
The influence of thermal regime on gasoline direct injection engine performance and emissions
 IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS The influence of thermal regime on gasoline direct injection engine performance and emissions To cite this article: C I Leahu
IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS The influence of thermal regime on gasoline direct injection engine performance and emissions To cite this article: C I Leahu
Simple Demonstration of the Seebeck Effect
 Simple Demonstration of the Seebeck Effect Arman Molki The Petroleum Institute, Abu Dhabi, United Arab Emirates amolki@pi.ac.ae Abstract In this article we propose a simple and low-cost experimental set-up
Simple Demonstration of the Seebeck Effect Arman Molki The Petroleum Institute, Abu Dhabi, United Arab Emirates amolki@pi.ac.ae Abstract In this article we propose a simple and low-cost experimental set-up
PEP Review HIGH-PURITY ISOBUTYLENE PRODUCTION BY MTBE CRACKING By Sumod Kalakkunnath (December 2012)
 PEP Review 2012-06 HIGH-PURITY ISOBUTYLENE PRODUCTION BY MTBE CRACKING By Sumod Kalakkunnath (December 2012) ABSTRACT This Review presents a technoeconomic evaluation of an isobutylene from methyl tertiary
PEP Review 2012-06 HIGH-PURITY ISOBUTYLENE PRODUCTION BY MTBE CRACKING By Sumod Kalakkunnath (December 2012) ABSTRACT This Review presents a technoeconomic evaluation of an isobutylene from methyl tertiary
Process Instrumentation ERT 213 Process Flow Diagram (PFD)
 Process Instrumentation ERT 213 Process Flow Diagram (PFD) Miss Hanna Ilyani Zulhaimi Outline Process diagram/flowsheeting Block Flow Diagram Process Flow Diagram Introduction to Microsoft Visio Learning
Process Instrumentation ERT 213 Process Flow Diagram (PFD) Miss Hanna Ilyani Zulhaimi Outline Process diagram/flowsheeting Block Flow Diagram Process Flow Diagram Introduction to Microsoft Visio Learning
CHAPTER 2 REFINERY FEED STREAMS: STREAMS FROM THE ATMOSPHERIC AND VACUUM TOWERS
 CHAPTER 2 REFINERY FEED STREAMS: STREAMS FROM THE ATMOSPHERIC AND VACUUM TOWERS About This Chapter The previous chapter introduced crude oil as a mixture of compounds. The characteristics of these compounds
CHAPTER 2 REFINERY FEED STREAMS: STREAMS FROM THE ATMOSPHERIC AND VACUUM TOWERS About This Chapter The previous chapter introduced crude oil as a mixture of compounds. The characteristics of these compounds
Reliable. Efficient. Economical. Distillation Technology ENGINEERING - EQUIPMENT - TURNKEY SYSTEMS
 TM Economical Efficient Reliable Distillation Technology ENGINEERING - EQUIPMENT - TURNKEY SYSTEMS DISTILLATION TECHNOLOGY Distillation is by far the most important separation process in the petroleum
TM Economical Efficient Reliable Distillation Technology ENGINEERING - EQUIPMENT - TURNKEY SYSTEMS DISTILLATION TECHNOLOGY Distillation is by far the most important separation process in the petroleum
Trouble: Flow Controller is unstable!
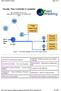 http://chem.engr.utc.edu/green-engineering/trouble-flow-controller.htm Page 1 of 1 By Jim Henry, Ph.D, P.E. University of Tennessee at Chattanooga 2004 Figure 1. Schematic diagram of the Filter Wash System
http://chem.engr.utc.edu/green-engineering/trouble-flow-controller.htm Page 1 of 1 By Jim Henry, Ph.D, P.E. University of Tennessee at Chattanooga 2004 Figure 1. Schematic diagram of the Filter Wash System
Biodiesel Unit Lesson 2
 Terminal Objective 2: produce biodiesel Biodiesel Unit Lesson 2 Performance Objective 2: Given unused cooking oil, necessary equipment and chemicals, produce biodiesel that is free of soap, dry, and ready
Terminal Objective 2: produce biodiesel Biodiesel Unit Lesson 2 Performance Objective 2: Given unused cooking oil, necessary equipment and chemicals, produce biodiesel that is free of soap, dry, and ready
Report. Refining Report. heat removal, lower crude preheat temperature,
 Delayed coker FCC feed hydrotreater FCCU Crude unit Hydrotreater Hydrotreater P r o c e s s i n g Better fractionation hikes yields, hydrotreater run lengths Scott Golden Process Consulting Services Houston
Delayed coker FCC feed hydrotreater FCCU Crude unit Hydrotreater Hydrotreater P r o c e s s i n g Better fractionation hikes yields, hydrotreater run lengths Scott Golden Process Consulting Services Houston
Crude Distillation Chapter 4
 Crude Distillation Chapter 4 Gases Gas Sat Gas Plant Polymerization LPG Sulfur Plant Sulfur Alkyl Feed Alkylation Butanes Fuel Gas LPG Gas Separation & Stabilizer Light Naphtha Heavy Naphtha Isomerization
Crude Distillation Chapter 4 Gases Gas Sat Gas Plant Polymerization LPG Sulfur Plant Sulfur Alkyl Feed Alkylation Butanes Fuel Gas LPG Gas Separation & Stabilizer Light Naphtha Heavy Naphtha Isomerization
ENGINEERING STANDARD FOR PROCESS DESIGN OF LPG RECOVERY AND SPLITTER UNITS ORIGINAL EDITION DEC. 1997
 ENGINEERING STANDARD FOR PROCESS DESIGN OF LPG RECOVERY AND SPLITTER UNITS ORIGINAL EDITION DEC. 1997 This standard specification is reviewed and updated by the relevant technical committee on Dec. 2002.
ENGINEERING STANDARD FOR PROCESS DESIGN OF LPG RECOVERY AND SPLITTER UNITS ORIGINAL EDITION DEC. 1997 This standard specification is reviewed and updated by the relevant technical committee on Dec. 2002.
correlated to the Virginia Standards of Learning, Grade 6
 correlated to the Virginia Standards of Learning, Grade 6 Standards to Content Report McDougal Littell Math, Course 1 2007 correlated to the Virginia Standards of Standards: Virginia Standards of Number
correlated to the Virginia Standards of Learning, Grade 6 Standards to Content Report McDougal Littell Math, Course 1 2007 correlated to the Virginia Standards of Standards: Virginia Standards of Number
March 7, 1967 J. F. G. ELLIS 3,308,060
 March 7, 1967 J. F. G. ELLIS PETROLEUM DISTILLATION Filed Jan. 28, 1965 NVENTOR. JOHN FRANCIS GRIFFITH ELLS BY MORGAN, FINNEGAN, DURHAM 8, PINE ATTORNEYs United States Patent Office Patented Mar. 7, 1967
March 7, 1967 J. F. G. ELLIS PETROLEUM DISTILLATION Filed Jan. 28, 1965 NVENTOR. JOHN FRANCIS GRIFFITH ELLS BY MORGAN, FINNEGAN, DURHAM 8, PINE ATTORNEYs United States Patent Office Patented Mar. 7, 1967
EXPERIMENTAL INVESTIGATION OF THERMAL PERFORMANCE OF PETROL ENGINE USING FUEL CATALYST
 EXPERIMENTAL INVESTIGATION OF THERMAL PERFORMANCE OF PETROL ENGINE USING FUEL CATALYST Sagar.A.Patil 1, Priyanka.V.Kadam 2, Mangesh.S.Yeolekar 3, Sandip.B.Sonawane 4 1 Student (Final Year), Department
EXPERIMENTAL INVESTIGATION OF THERMAL PERFORMANCE OF PETROL ENGINE USING FUEL CATALYST Sagar.A.Patil 1, Priyanka.V.Kadam 2, Mangesh.S.Yeolekar 3, Sandip.B.Sonawane 4 1 Student (Final Year), Department
PVP Field Calibration and Accuracy of Torque Wrenches. Proceedings of ASME PVP ASME Pressure Vessel and Piping Conference PVP2011-
 Proceedings of ASME PVP2011 2011 ASME Pressure Vessel and Piping Conference Proceedings of the ASME 2011 Pressure Vessels July 17-21, & Piping 2011, Division Baltimore, Conference Maryland PVP2011 July
Proceedings of ASME PVP2011 2011 ASME Pressure Vessel and Piping Conference Proceedings of the ASME 2011 Pressure Vessels July 17-21, & Piping 2011, Division Baltimore, Conference Maryland PVP2011 July
Thermal Analysis of Shell and Tube Heat Exchanger Using Different Fin Cross Section
 Thermal Analysis of Shell and Tube Heat Exchanger Using Different Fin Cross Section J. Heeraman M.Tech -Thermal Engineering Department of Mechanical Engineering Ellenki College of Engineering & Technology
Thermal Analysis of Shell and Tube Heat Exchanger Using Different Fin Cross Section J. Heeraman M.Tech -Thermal Engineering Department of Mechanical Engineering Ellenki College of Engineering & Technology
Comparing Percentages of Iditarod Finishers
 Comparing Percentages of Iditarod Finishers Developed by: Brian Hickox, 2019 Iditarod Teacher on the Trail Discipline / Subject: Math Topic: Analyze and Solve Percent Problems Grade Level: 6, 7, 8 Resources
Comparing Percentages of Iditarod Finishers Developed by: Brian Hickox, 2019 Iditarod Teacher on the Trail Discipline / Subject: Math Topic: Analyze and Solve Percent Problems Grade Level: 6, 7, 8 Resources
STUDIES ON FUSHUN SHALE OIL FURFURAL REFINING
 Oil Shale, 2011, Vol. 28, No. 3, pp. 372 379 ISSN 0208-189X doi: 10.3176/oil.2011.3.02 2011 Estonian Academy Publishers STUDIES ON FUSHUN SHALE OIL FURFURAL REFINING G. X. LI, D. Y. HAN *, Z. B. CAO, M.
Oil Shale, 2011, Vol. 28, No. 3, pp. 372 379 ISSN 0208-189X doi: 10.3176/oil.2011.3.02 2011 Estonian Academy Publishers STUDIES ON FUSHUN SHALE OIL FURFURAL REFINING G. X. LI, D. Y. HAN *, Z. B. CAO, M.
Technical Notes. Introduction. LED Efficacy. Light Output and Efficacy. Issue 4 April The general equation for light output can be written as:
 Technical Notes Lighting Efficiency and Product Design Optimization This Technical Note discusses lighting product design from an energy and efficiency perspective. It is intended to help manufacturers
Technical Notes Lighting Efficiency and Product Design Optimization This Technical Note discusses lighting product design from an energy and efficiency perspective. It is intended to help manufacturers
EXPERIMENTAL INVESTIGATION OF FOUR STROKE SINGLE CYLINDER DIESEL ENGINE WITH OXYGENATED FUEL ADDITIVES
 EXPERIMENTAL INVESTIGATION OF FOUR STROKE SINGLE CYLINDER DIESEL ENGINE WITH OXYGENATED FUEL ADDITIVES 1 Bhavin Mehta, 2 Hardik B. Patel 1,2 harotar University of Science & Technology, Changa, Gujarat,
EXPERIMENTAL INVESTIGATION OF FOUR STROKE SINGLE CYLINDER DIESEL ENGINE WITH OXYGENATED FUEL ADDITIVES 1 Bhavin Mehta, 2 Hardik B. Patel 1,2 harotar University of Science & Technology, Changa, Gujarat,
Hydrocracking of atmospheric distillable residue of Mongolian oil
 Hydrocracking of atmospheric distillable residue of Mongolian oil Ts.Tugsuu 1, Sugimoto Yoshikazu 2, B.Enkhsaruul 1, D.Monkhoobor 1 1 School of Chemistry and Chemical Engineering, NUM, PO Box-46/574, Ulaanbaatar
Hydrocracking of atmospheric distillable residue of Mongolian oil Ts.Tugsuu 1, Sugimoto Yoshikazu 2, B.Enkhsaruul 1, D.Monkhoobor 1 1 School of Chemistry and Chemical Engineering, NUM, PO Box-46/574, Ulaanbaatar
Frequency Response Curve For Forced Vibration under Different Damping for Steel Beam
 Frequency Response Curve For Forced Vibration under Different Damping for Steel Beam Miss. Kachare Savita, Miss. Mhaske Priyanka, Miss. Phate Jyoti Lecturer, Dept of Mechanical Engg., Vishwabharati Academy
Frequency Response Curve For Forced Vibration under Different Damping for Steel Beam Miss. Kachare Savita, Miss. Mhaske Priyanka, Miss. Phate Jyoti Lecturer, Dept of Mechanical Engg., Vishwabharati Academy
Components of Hydronic Systems
 Valve and Actuator Manual 977 Hydronic System Basics Section Engineering Bulletin H111 Issue Date 0789 Components of Hydronic Systems The performance of a hydronic system depends upon many factors. Because
Valve and Actuator Manual 977 Hydronic System Basics Section Engineering Bulletin H111 Issue Date 0789 Components of Hydronic Systems The performance of a hydronic system depends upon many factors. Because
COMPUTATIONAL FLOW MODEL OF WESTFALL'S 2900 MIXER TO BE USED BY CNRL FOR BITUMEN VISCOSITY CONTROL Report R0. By Kimbal A.
 COMPUTATIONAL FLOW MODEL OF WESTFALL'S 2900 MIXER TO BE USED BY CNRL FOR BITUMEN VISCOSITY CONTROL Report 412509-1R0 By Kimbal A. Hall, PE Submitted to: WESTFALL MANUFACTURING COMPANY May 2012 ALDEN RESEARCH
COMPUTATIONAL FLOW MODEL OF WESTFALL'S 2900 MIXER TO BE USED BY CNRL FOR BITUMEN VISCOSITY CONTROL Report 412509-1R0 By Kimbal A. Hall, PE Submitted to: WESTFALL MANUFACTURING COMPANY May 2012 ALDEN RESEARCH
CFD Analysis of Oil Discharge Rate in Rotary Compressor
 Purdue University Purdue e-pubs International Compressor Engineering Conference School of Mechanical Engineering CFD Analysis of Oil Discharge Rate in Rotary Compressor Liying Deng haitunsai@.com Shebing
Purdue University Purdue e-pubs International Compressor Engineering Conference School of Mechanical Engineering CFD Analysis of Oil Discharge Rate in Rotary Compressor Liying Deng haitunsai@.com Shebing
Petroleum Refining Fourth Year Dr.Aysar T. Jarullah
 Catalytic Operations Fluidized Catalytic Cracking The fluidized catalytic cracking (FCC) unit is the heart of the refinery and is where heavy low-value petroleum stream such as vacuum gas oil (VGO) is
Catalytic Operations Fluidized Catalytic Cracking The fluidized catalytic cracking (FCC) unit is the heart of the refinery and is where heavy low-value petroleum stream such as vacuum gas oil (VGO) is
A Novel Device to Measure Instantaneous Swept Volume of Internal Combustion Engines
 Global Journal of Researches in Engineering Vol. 10 Issue 7 (Ver1.0), December 2010 P a g e 47 A Novel Device to Measure Instantaneous Swept Volume of Internal Combustion Engines MURUGAN. R. GJRE -A Classification
Global Journal of Researches in Engineering Vol. 10 Issue 7 (Ver1.0), December 2010 P a g e 47 A Novel Device to Measure Instantaneous Swept Volume of Internal Combustion Engines MURUGAN. R. GJRE -A Classification
BIODIESEL. Lesson 4a Test biodiesel in an oil lamp. Developed by Engineering and Technology Curriculum Team 2008
 BIODIESEL Lesson 4a Test biodiesel in an oil lamp In a laboratory setting, test biodiesel in an oil lamp by estimating calories and Btu. define density, energy density, calories and Btu calculate density,
BIODIESEL Lesson 4a Test biodiesel in an oil lamp In a laboratory setting, test biodiesel in an oil lamp by estimating calories and Btu. define density, energy density, calories and Btu calculate density,
Meeting product specifications
 Optimisation of a diesel hydrotreating unit A model based on operating data is used to meet sulphur product specifications at lower DHT reactor temperatures with longer catalyst life Jose Bird Valero Energy
Optimisation of a diesel hydrotreating unit A model based on operating data is used to meet sulphur product specifications at lower DHT reactor temperatures with longer catalyst life Jose Bird Valero Energy
PEP Review HIGH-PURITY ISOBUTYLENE FROM T-BUTANOL BY LYONDELLBASELL PROCESS By Sumod Kalakkunnath (February 2013)
 PEP Review 2013-06 HIGH-PURITY ISOBUTYLENE FROM T-BUTANOL BY LYONDELLBASELL PROCESS By Sumod Kalakkunnath (February 2013) ABSTRACT This Review presents a technoeconomic evaluation of an isobutylene from
PEP Review 2013-06 HIGH-PURITY ISOBUTYLENE FROM T-BUTANOL BY LYONDELLBASELL PROCESS By Sumod Kalakkunnath (February 2013) ABSTRACT This Review presents a technoeconomic evaluation of an isobutylene from
Measurement of induction motor characteristics
 Measurement of induction motor characteristics ES163 Electrical and Electronic Systems MR TJ KENNAUGH School of Engineering, University of Warwick 27/01/01 Summary The aim of the laboratory is to increase
Measurement of induction motor characteristics ES163 Electrical and Electronic Systems MR TJ KENNAUGH School of Engineering, University of Warwick 27/01/01 Summary The aim of the laboratory is to increase
Battery Capacity Versus Discharge Rate
 Exercise 2 Battery Capacity Versus Discharge Rate EXERCISE OBJECTIVE When you have completed this exercise, you will be familiar with the effects of the discharge rate and battery temperature on the capacity
Exercise 2 Battery Capacity Versus Discharge Rate EXERCISE OBJECTIVE When you have completed this exercise, you will be familiar with the effects of the discharge rate and battery temperature on the capacity
International Journal of Mechanical And Production Engineering, ISSN: , GRADE ANALYSIS OF PYROLYSIS OIL BY STEP DISTILLATION
 GRADE ANALYSIS OF PYROLYSIS OIL BY STEP DISTILLATION 1 AKSHAY S. NAKHATE, 2 ONKAR SAHASRABUDHE ¹Student,M.E. Thermal engineering, ²Assistant professor, Department of Mechanical Engineering, Pillai Institute
GRADE ANALYSIS OF PYROLYSIS OIL BY STEP DISTILLATION 1 AKSHAY S. NAKHATE, 2 ONKAR SAHASRABUDHE ¹Student,M.E. Thermal engineering, ²Assistant professor, Department of Mechanical Engineering, Pillai Institute
High Throughput Mineral Oil Analysis (Hydrocarbon Oil Index) by GC-FID Using the Agilent Low Thermal Mass (LTM) System
 High Throughput Mineral Oil Analysis (Hydrocarbon Oil Index) by GC-FID Using the Agilent Low Thermal Mass (LTM) System Application Note Authors Frank David Research Institute for Chromatography, Pres.
High Throughput Mineral Oil Analysis (Hydrocarbon Oil Index) by GC-FID Using the Agilent Low Thermal Mass (LTM) System Application Note Authors Frank David Research Institute for Chromatography, Pres.
Simulation and Optimization of H 2 S Expulsion from Crude Oil with the Use of Equilibrium Model
 2011 International Conference on Chemistry and Chemical Process IPCBEE vol.10 (2011) (2011) IACSIT Press, Singapore Simulation and Optimization of H 2 S Expulsion from Crude Oil with the Use of Equilibrium
2011 International Conference on Chemistry and Chemical Process IPCBEE vol.10 (2011) (2011) IACSIT Press, Singapore Simulation and Optimization of H 2 S Expulsion from Crude Oil with the Use of Equilibrium
Filtertechnik Filtration, Purification & Separation Solutions
 Titration kit for biodiesel production Filtertechnik Filtration, Purification & Separation Solutions Using this kit will enable you to accurately determine the amount of Free Fatty Acid (FFA) in your vegetable
Titration kit for biodiesel production Filtertechnik Filtration, Purification & Separation Solutions Using this kit will enable you to accurately determine the amount of Free Fatty Acid (FFA) in your vegetable
INTERNATIONAL JOURNAL OF APPLIED ENGINEERING RESEARCH, DINDIGUL Volume 1, No 4, 2011
 Numerical modal analysis of Howell Bunger valve using FEM method Farid Vakili Tahami, Mohammad Zehsaz, Mohammad Ali Saeimi Sadigh, Amin Paykani Department of Mechanical Engineering, University of Tabriz,
Numerical modal analysis of Howell Bunger valve using FEM method Farid Vakili Tahami, Mohammad Zehsaz, Mohammad Ali Saeimi Sadigh, Amin Paykani Department of Mechanical Engineering, University of Tabriz,
Laboratory 2 Electronics Engineering 1270
 Laboratory 2 Electronics Engineering 1270 DC Test Equipment Purpose: This lab will introduce many of the fundamental test equipment and procedures used for verifying the operations of electrical circuits.
Laboratory 2 Electronics Engineering 1270 DC Test Equipment Purpose: This lab will introduce many of the fundamental test equipment and procedures used for verifying the operations of electrical circuits.
REPORT SYNTHETIC AND MINERAL CRUDE OILS COMPATIBILITY STUDY
 REPORT SYNTHETIC AND MINERAL CRUDE OILS COMPATIBILITY STUDY Moscow - 2013 TABLE OF CONTENTS Page Introduction... 3 1. Selecting and adapting the existing methods of compatibility study for mixed synthetic
REPORT SYNTHETIC AND MINERAL CRUDE OILS COMPATIBILITY STUDY Moscow - 2013 TABLE OF CONTENTS Page Introduction... 3 1. Selecting and adapting the existing methods of compatibility study for mixed synthetic
DESIGN AND ANALYSIS OF CAR RADIATOR BY FINITE ELEMENT METHOD
 DESIGN AND ANALYSIS OF CAR RADIATOR BY FINITE ELEMENT METHOD Prof. V. C. Pathade 1, Sagar R. Satpute 2, Mayur G. Lajurkar 3, Gopal R. Pancheshwar 4 Tushar K. Karluke 5, Niranjan H. Singitvar 6 1 Assistant
DESIGN AND ANALYSIS OF CAR RADIATOR BY FINITE ELEMENT METHOD Prof. V. C. Pathade 1, Sagar R. Satpute 2, Mayur G. Lajurkar 3, Gopal R. Pancheshwar 4 Tushar K. Karluke 5, Niranjan H. Singitvar 6 1 Assistant
